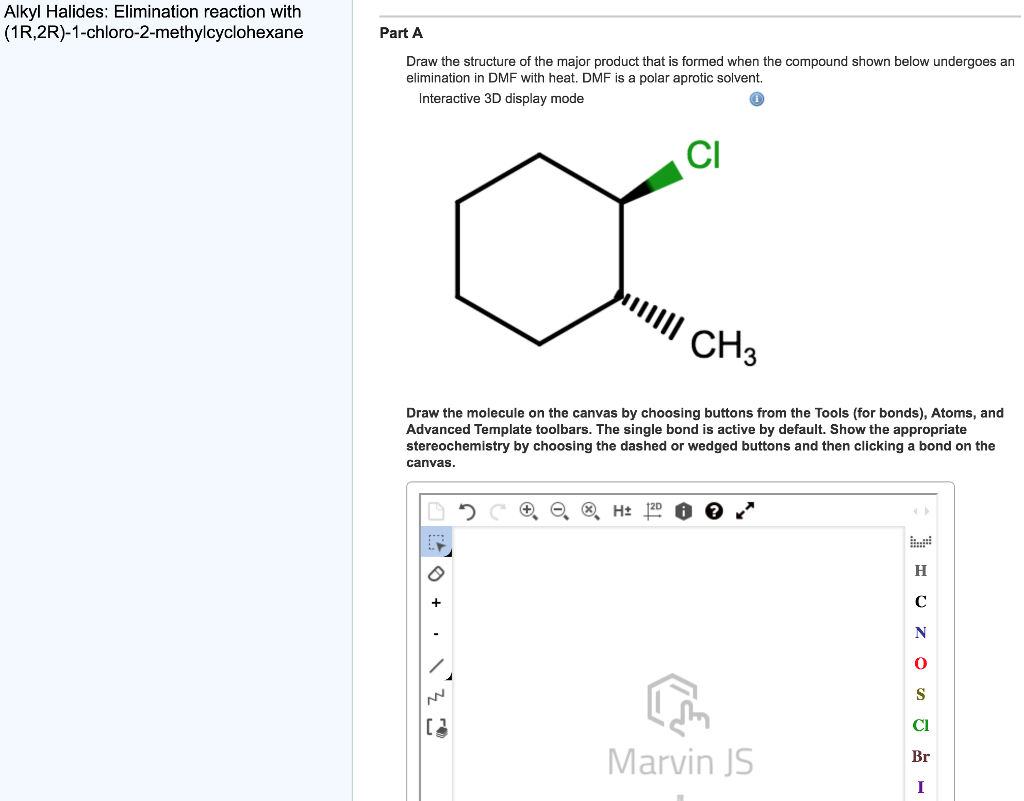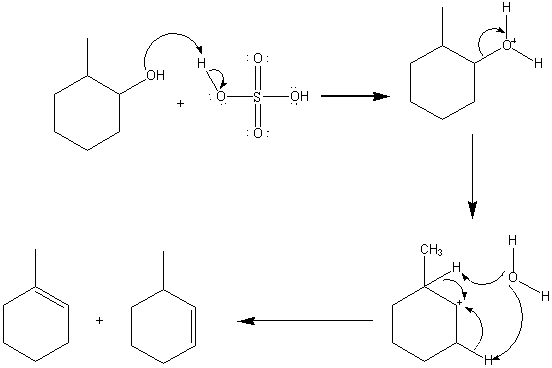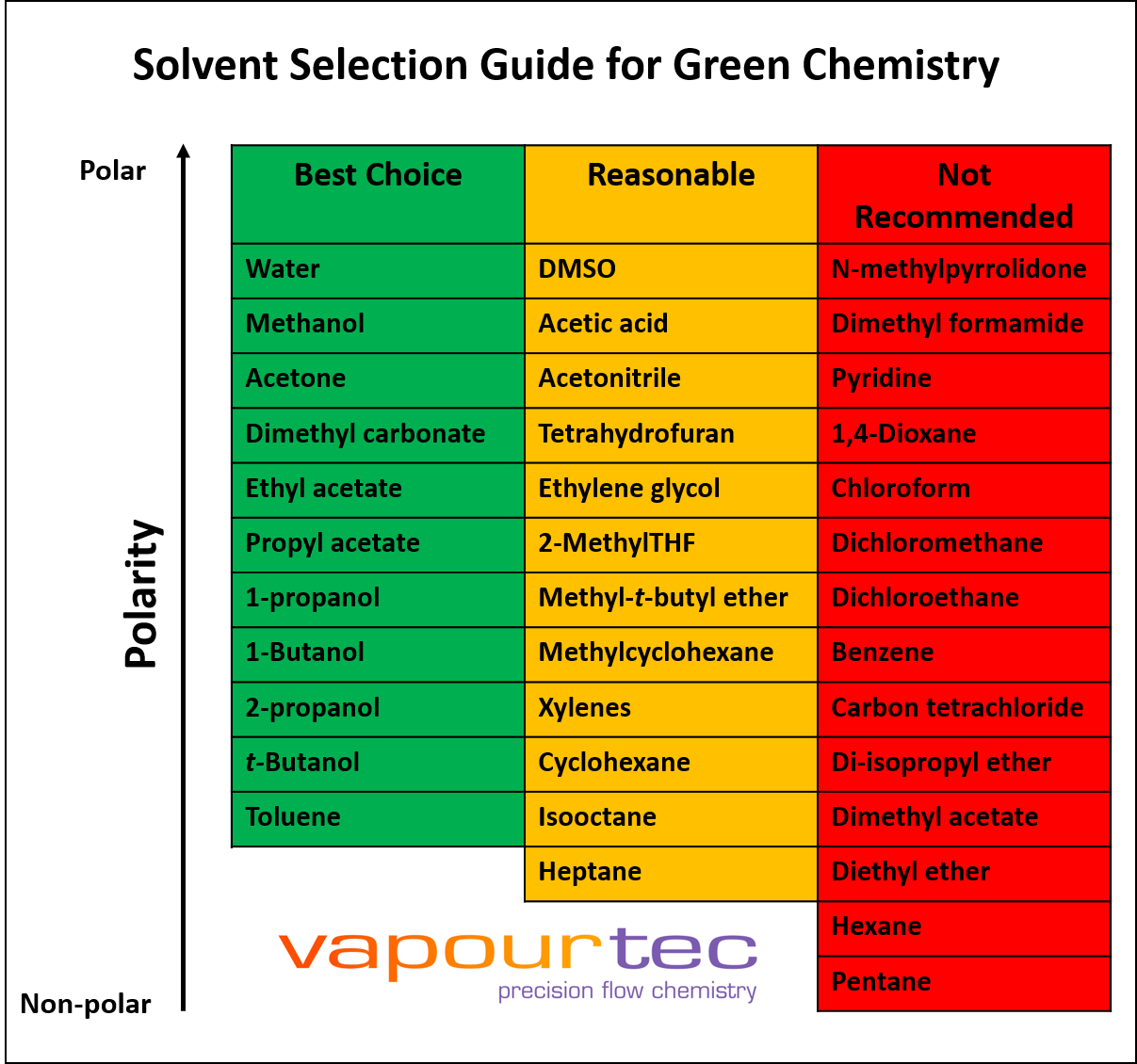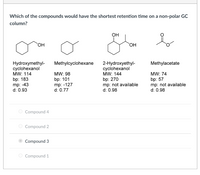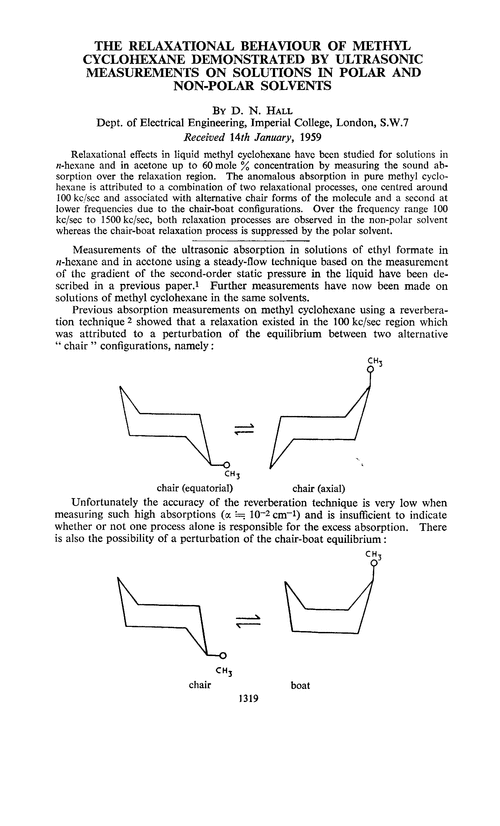
The relaxational behaviour of methyl cyclohexane demonstrated by ultrasonic measurements on solutions in polar and non-polar solvents - Transactions of the Faraday Society (RSC Publishing)
Inorganics | Free Full-Text | Study of Cyclohexane and Methylcyclohexane Functionalization Promoted by Manganese(III) Compounds

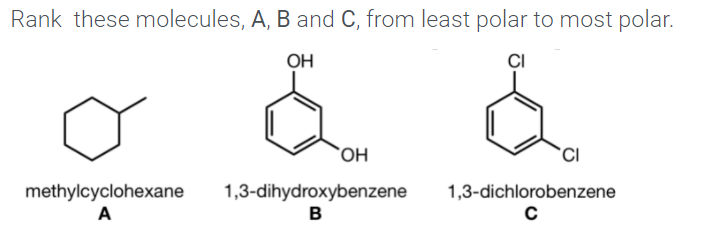
Excited‐State Symmetry Breaking in an Aza‐Nanographene Dye - Bardi - 2019 - Chemistry – A European Journal - Wiley Online Library

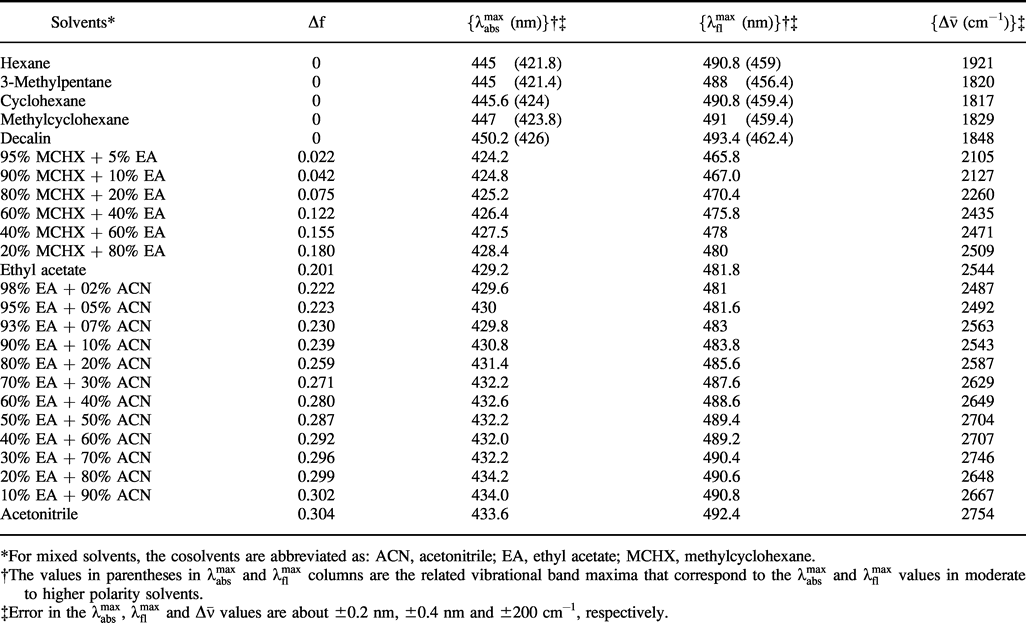
Effective Polarity of Frozen Solvent Glasses in the Vicinity of Dipolar Solutes | Journal of the American Chemical Society

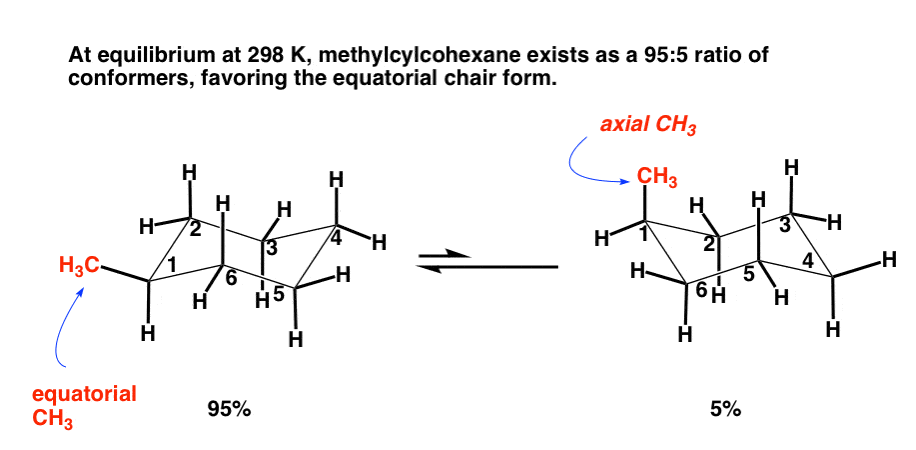
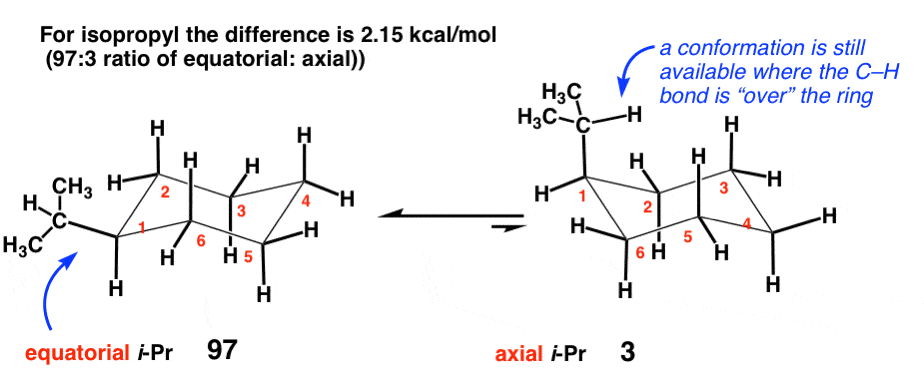
One of the two chair structures of cis-1-chloro-3-methylcyclohexane is more stable than the other by 15.5 \frac{kJ}{mol} (3.7 \frac{kcal}{mol}). Which is it? What is the energy cost of a 1,3-diaxial interaction between